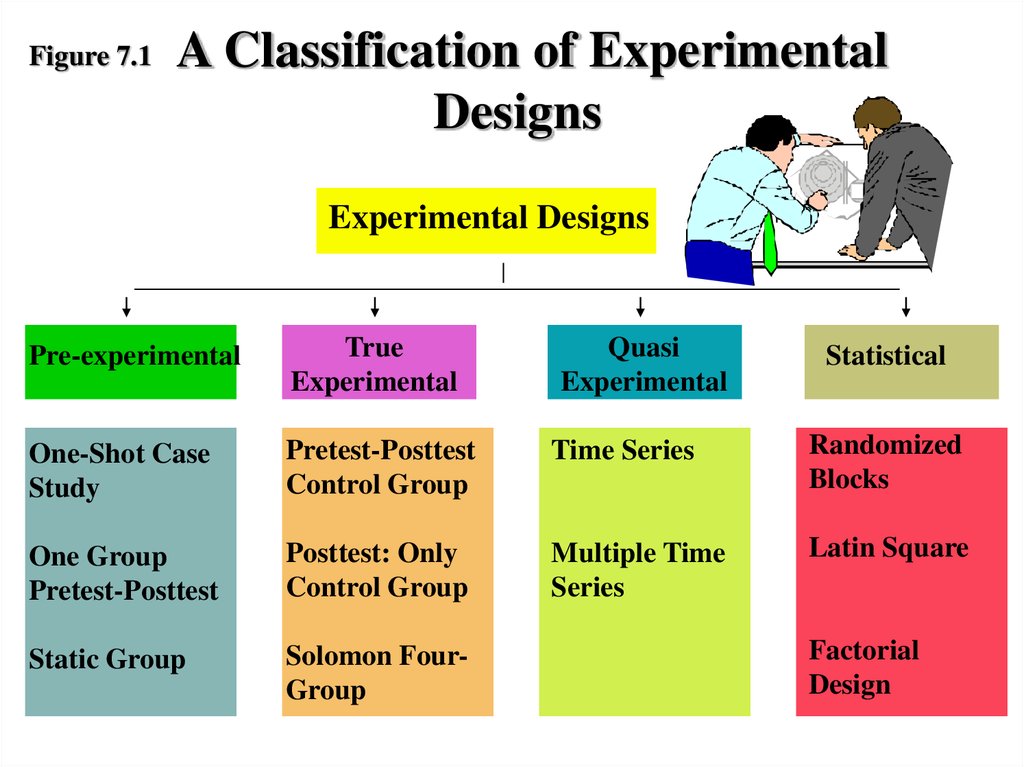 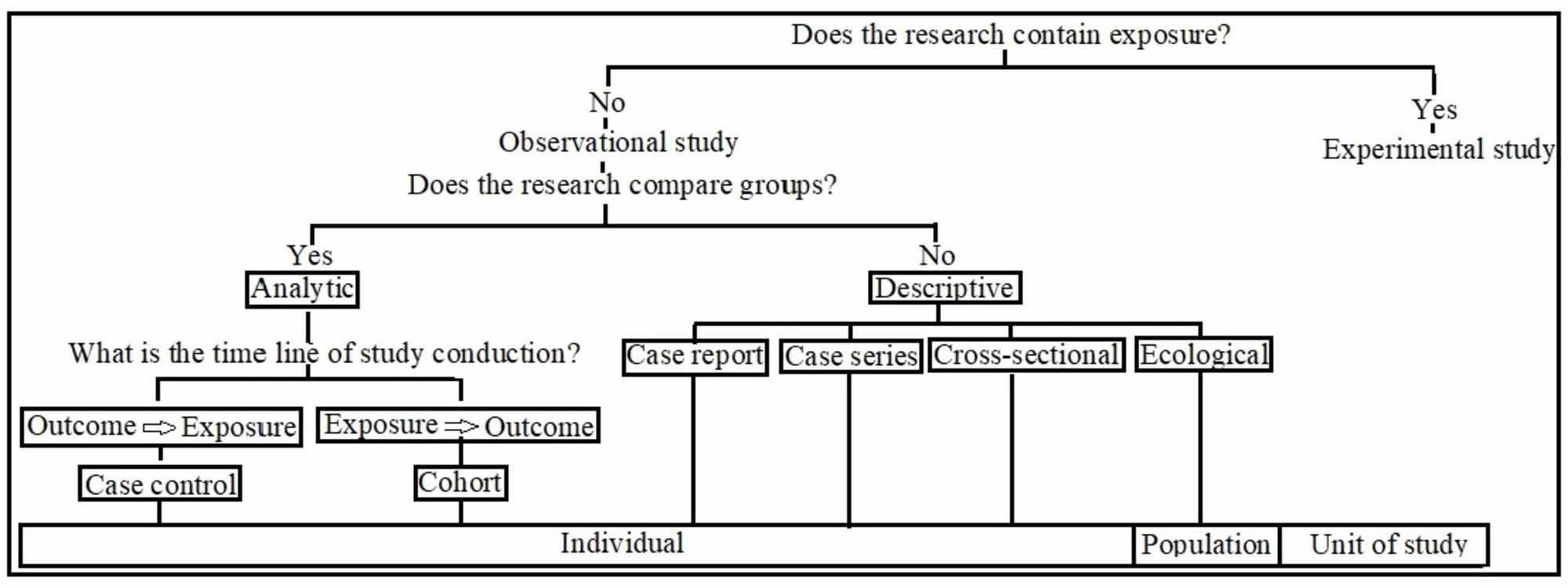
The study ends with a discussion of study endpoints, data management, and statistical analysis. The hope is that the overview provided here, with the aid of specific references provided, aids the researcher in designing and conducting a clinical research study. Good experimental design requires that the researchers and the study personnel take great care when performing study-related tasks from the moment the hypothesis is generated to the time of final analysis. A well designed, executed, and analyzed clinical research study on a sample, or, that is, a study with good experimental design, gives the researchers the best chance of obtaining study results applicable to the population from which the samples are taken. The study follows a discussion of one of the more important aspects of a good study design: sample. Further, it reviews the various study design options, followed by the definition of bias and how to reduce or minimize biases in the conduct of the study. It discusses the first important step: developing hypotheses to be studied. The chapter describes the major components of good experimental design. Joseph Massaro, in Clinical and Translational Science, 2009 Publisher Summary
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
